Category
- NEW PRODUCTS
- CUSTOMER SERVICES
- Custom made glass products
- Custom made and Modified Screen Printed Electrodes
- Sensors and electrodes
- Cables and connectors
- Cell
- Spectro, Photo, Raman – electrochemical cells
- Membrane Capacitive Deionization configurable cell
- NREL High-Pressure Low-Temperature Electrolysis Cell
- High-Pressure (25 bar) Single-Compartment Electrochemical Cell
- Stirrers
- Pumps
- Kits & Sets
- Minithermostat
- Modular PEM Electrolyzer Test Station
- Measurements of battery and supercapacitor materials
- Potentiostats
- Manual Screen Printer
- Accessories
- Discounted SPEs (at a reduced price with visual defects/inconsistancies, but fully functional)
Membrane Capacitive Deionization configurable cell (redox.me)
Membrane Capacitive Deionization (MCDI) configurable cell has been designed to conduct research on removal of charged ionic species from aqueous or organic solutions (i.e., Desalination/Demineralization) via electrostatic (i.e., non-Faradaic) or electrochemical (redox) interactions. The MCDI cell contains two graphite current collectors that can serve as polarization electrodes. However, if the electrode material is the subject of research, it should be applied to an additional current collector, such as graphite paper or metal felt (not included in the product). Different electrode materials can be installed on both sides of the cell. If the thickness of these electrodes exceeds 250 micrometers, different gaskets are required. In such a case, please contact us for a solution.
The cell is designed in such a way that replacing the electrodes does not require removing the membranes or flow fields. It is enough to unscrew the plungers on both sides and replace the electrodes. In the standard configuration, the liquid in the main channel and side channels flows through the flow field cut from PEEK. However, these can be replaced with a porous material such as felt or a battery spacer, which will allow fluid flow. If work with materials of dimensions different from those listed in the specification is required, we can supply seals of different thicknesses or customize the cell. The cell allows for the installation of one or two ion-exchange membranes (not included in the setup). Their thickness is not critical, and it is not necessary to adjust the thickness of the membrane gaskets pressing them. The cell elements are constructed with inert materials to the sample (PEEK). It well fits aqueous (FKM gaskets and O-rings) and organic solvent (FFKM gaskets and O-rings) electrolyte requirements. The construction of the cell is gas-tight.
Application note:
MCDI cell can be configured to allow the following cell architectures:
- Flow-by CDI consisting of: (i) two porous carbon or metal based current collectors coated with capacitive (e. non-Faradaic) material, and (ii) a main flow field enabling the feed water to be transported between electrodes. In this configuration, side flow fields and membranes are not installed.
- Membrane CDI consisting of: (i) two porous carbon or metal based current collectors coated with capacitive (e. non-Faradaic) material, (ii) two ion-exchange membranes (cation exchange membrane and anion exchange membrane) separating electrodes from the main flow field, and (iii) a main flow field enabling the feed water to be transported between electrodes. In this configuration, side flow fields are typically not installed. However, there are cases where installing both side flow fields is justified. All the graphics included in the product page refer to that configuration.
- Inverted CDI consisting of: (i) two porous carbon or metal based current collectors coated with capacitive (e. non-Faradaic) material where anode is treated for net negative surface charge and a cathode is treated for net positive surface charge, and (ii) a main flow field enabling the feed water to be transported between electrodes. In this configuration, side flow fields and membranes are not installed.
- Flow-electrode CDI consisting of: (i) two porous carbon or metal based current collectors with flowing electrodes made of capacitive (e. non-Faradaic) carbon suspension, (ii) two ion-exchange membranes (cation exchange membrane and anion exchange membrane) separating electrodes from the flow chamber, (iii) a main flow field enabling the feed water to be transported between electrodes, and (iv) two side flow fields for liquid electrodes. All the graphics included in the product page refer to that configuration.
- Hybrid CDI consisting of: (i) a Faradaic (e. battery) electrode for cation adsorption/desorption, (ii) a capacitive (i.e. non-Faradaic) electrode for anion adsorption/desorption, (iii) an anion exchange membrane placed adjacent to the capacitive electrode, and (iv) a main flow field enabling the feed water to be transported between electrodes. In this configuration, side flow fields and a cation-exchange membrane are not installed.
- Cation intercalation desalination consisting of: (i) two porous carbon or metal based current collectors coated with Faradaic cation intercalation materials, (ii) an anion exchange membrane separating electrodes, and (iii) a main flow field enabling the feed water to be transported between electrodes. In this configuration, one side flow field and cation-exchange membrane are not installed.
- Desalination battery consisting of: (i) two porous carbon or metal based current collectors coated with redox (e. Faradaic) material (one for cation adsorption/desorption and the other for anion adsorption/desorption), and (ii) a main flow field enabling the feed water to be transported between electrodes. In this configuration, side flow fields and membranes are not installed.
Specification:
tubing size: 4 mm OD
fitting type: push-in, M5 male
electrode size: 60 mm x 60 mm (36 cm2)
recommended total electrode thickness: 200-250 µm
membrane size: 70 mm x 85 mm
maximum operating pressure: 20 bar
maximum operating temperature 150 ºC
Intrastat data:
HS Code: 90278080
Country of Origin: Sweden
NET weight: 1300 g
Product includes:
2 x stand, anodized aluminum
2 x plunger holder, SS 316L
2 x PEEK plunger
2 x tantalum current collector
2 x graphite current collector
1 x threaded end plate, SS 316L
1 x unthreaded end plate, SS 316L
1 x PEEK outer cell body
1 x PEEK inner cell body
1 x set of fittings
2 x female banana connectors, 4 mm dia.
1 x PEEK main flow field, 0.5 mm thick
2 x PEEK side flow field, 0.5 mm thick
1 x set of gaskets (FKM or FFKM) including:
1 x main flow gasket, 0.5 mm thick
2 x membrane gasket, 0.25 mm thick
2 x side flow gasket, 0.5 mm thick
2 x electrode gasket, 0.25 mm thick
Select configuration
Electrolyte type: water-based / organic electrolyte
Related products
-
ZIVE SP1 – potentiostat/galvanostat/ZRA
Read more- Potentiostat/galvanostat/ZRA at affordable price
- Control voltage range: ±10V
- Control current range:9 ranges, 10nA~1A (10nA with gain)
Application - Battery
- Super capacitor
- Fuel cell
- Corrosion
- Sensor
- Solar cell
- Other Echem experiments
Features
- economical high quality potentiostat/galvanostat/impedance analyzer
- compact size with full functions
- ±10V@1Amp control range
- wide current ranges(1A to 10nA) for various applications
- built-in FRA : enables EIS tests by using software
- 14 EIS techniques capability including multisine
- capable of multitude of applications
– corrosion, general electrochemistry, sensor, battery, fuel cell,
supercapacitor, solar cell, etc. - bipolar pulse capability
- voltage pulse or current pulse charge/discharge test(GSM,CDMA etc.),
sine wave function for ripple simulation withenergysoftwarepackage
& pulse plating available - high speed data sampling time
– 2usec or 3usec depending on data point number - iR compensation and measurement
- 3 measurement/control voltage ranges &
9 measurement/control current ranges - internal 542,000 data point storage & continuing experiment regardless
of PC failure. - multichannel configuration available
- free software upgrade
Experimental Techniques
Basic techniques
- Potentiostatic
- Galvanostatic
- Double step potentiostatic
- Double step galvanostatic
- OCP measurement
- Potential sweep
- Current sweep
- Cyclic voltammetry
- Fast potential sweep
- Potentiostatic Ru measurement
- Galvanostatic Ru measurement
Advanced Software Package(Included)
- EIS software package(EISe)
– Potentiostat EIS
– Galvanostatic EIS
– Pseudo galvanostatic EIS
– OCP* EIS
– Potentiodynamic PEIS
– Galvanodynamic GEIS
– Poteniodynamic HFR
– Galvanodynamic HFR
– Potentiostatic HFR monitor
– Galvanostatic HFR monitor
– Multisine potentiostatic EIS
– Multisine galvanostatic EIS
– Intermittent potentiostatic EIS
– Intermittent galvanostatic EIS
(*) The system measures open circuit potential before each frequency
change and applies AC sine wave on this potential. - Corrosion software package(CORe)
– Tafel(Tafel experiment)
– Rp(Polarization resistance)
– RpEc trend
– PDYN(Potentiodynamic)
– CYPOL(Cyclic polarization resistance)
– GDYN(GalvanoDynamic)
– Reactivation
– Galvanic corrosion
– Potentiostatic ECN
– Galvanostatic ECN
– ZRA mode ECN - Energy software package(BATe)
a) Battery test technique
– CC/CV testforcycle life test of lithium battery
– CC/CC tet forcyclelifetestofNiCd or NiMH battery
– Discharging test
– EVS(electrochemical voltage spectroscopy)
– Variable scan rate CV
– Potentiostatic IV curve
– Galvanostatic IV curve
– Steady state CV
– PITT(Potentiostatic intermittent titration technique) test
– GITT(Galvanostatic intermittent titration technique) test
– Pulse mode is available for GSM & CDMA profile.
Pulse shape profile can be measured by user’s demand.
b) Control mode
– Charge : CC, CC-CV, pulse, sine wave
– Discharge : CC, CP, CR, pulse, sine wave
c) Cut-off condition
– Time, voltage, current, power, auxV etc.
– Various battery charge/discharge test is available including
pulse discharge for GSM, CDMA application - Electrochemical analysis software package(EASe)
a) Step techniques
– CA(Chronoamperometry)
– CC(Chronocoulometry)
– CP(Chronopotentiometry)
b) Sweep techniques
– LSV(Linear sweep voltammetry)
– SDV(Sampled DC voltammetry)
– Fast CV
– Fast LSV
c) Pulsed techniques
– DPV(Differential pulse voltammetry)
– SWV(Square wave voltammetry)
– DPA(Diff.pulse amperometry)
– NPV(Normal pulse voltammetry)
– RNPV(Reverse normal pulse voltammetry)
– DNPV(Differential normal pulse voltammetry)
-
SPE Connector.GC.1 for measurements in glass
Read moreThe connector enables the use of the biosensor or electrochemical sensor based on the substrates AC1, AC4 and TS1 in glass cell TC4, TC5, TC6 or TC7.
The connector enables measurement with screen printed electrodes with width 7.26 mm and standard contact pads pitch (2.54 mm).
It is compatible with other SPE´s that use the same distance between the contact pads (2.54 mm). The cone at the connector sensor side is NJ 8/10 which enables to use the connector with any standard chemical vessels.Termination: 2 mm Banana plugs
-
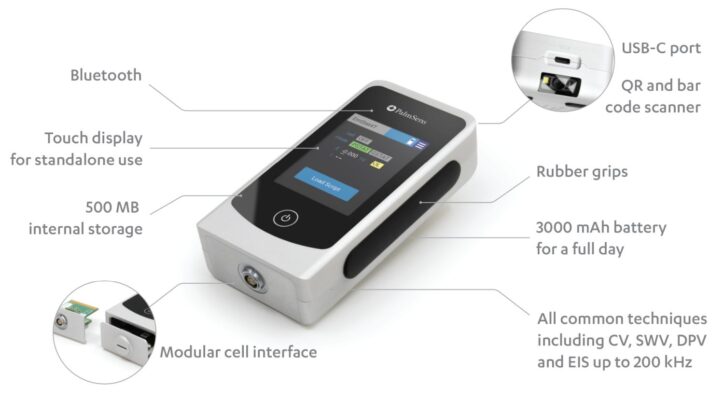
EmStat4T (Tailored electrochemistry through touch-controlled apps)
Read moreThe EmStat4T is a programmable handheld potentiostat with a touchscreen, which is ideal for sensor research and sensor-based applications. It offers two main modes of operation:
- Remote Control: where it functions as a conventional potentiostat, controlled directly by our PSTrace software for Windows or PStouch app for Android. These applications allow you to run measurements, view results, and perform data analysis.
- Standalone: where the instrument is controlled via its touch interface to run a wizard‑style app for electrochemical analysis. Compose custom apps easily using the Visual MethodSCRIPT Editor included in PSTrace for Windows. Apps eliminate the need for a computer, tablet, or smartphone. This makes the EmStat4T an ideal solution for point-of-care applications and field research such as environmental analysis or corrosion monitoring.
More information could be found tought link:
https://www.palmsens.com/emstat4t/
Tailored electrochemistry through touch-controlled apps
- Potential range ±3 V
- Max. current ±30 mA
- Supports common electrochemical techniques
- Standalone operation with MethodSCRIPT
- or remotely controlled by laptop or phone
- Bar/QR-code scanner
- Customizable cell interface with drop-detection for SPEs
Main Features
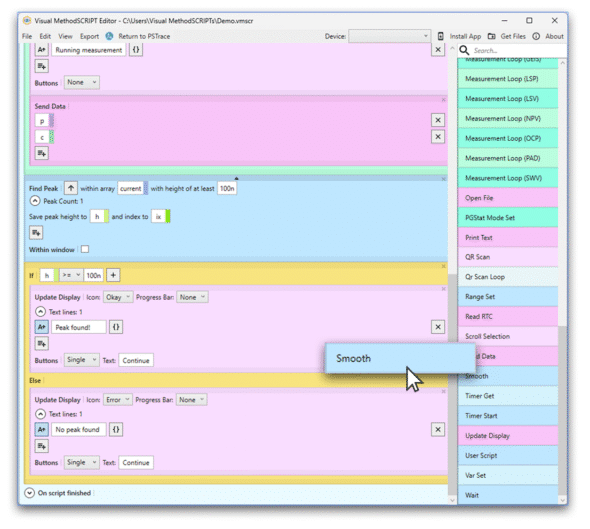 Visual MethodSCRIPT Editor
Visual MethodSCRIPT EditorCreate your EmStat4T apps
The powerful MethodSCRIPT™ language allows for easily creating your own applications to run on the EmStat4T. Compose apps using our Visual MethodSCRIPT Editor which generates the MethodSCRIPT for you.
Your measurements are safely stored

Internal Storage
The EmStat4T is equipped with 500 MB internal storage memory for storing your measurements. All internally stored measurements can be browsed and transferred back to the PC easily using the PSTrace software for Windows. Your data is always with your instrument wherever you take it.
-
Raman Electrochemical Flow Cell, active area: 3.5 cm2, volume: 4.5 mL (redox.me)
Read moreThis cell combines two classical analytical techniques such as electrochemistry and Raman spectroscopy, to obtain in-situ chemical information about the reactions taking place during an electrochemical experiment. It consists mainly of three elements: (i) sample holder with Tantalum (formerly Copper) foil serving as an electric contact to working electrode surface, (ii) chamber in which reference and counter electrodes are installed together with electrolyte inlet and outlet and (iii) lid which seals the cell and holds the Sapphire window. The sample consisting of a thin film of electrochemically active material deposited on a rigid or flexible substrate (working electrode) is loaded from the bottom via magnetic or screw mount. The counter or/and reference electrodes are mounted in a top casing (either 2-, or 3-electrode setup. The oval counter electrode made of e.g. Platinum wire assures uniform distribution of the field lines along the path to working electrode. During an experiment Raman laser is focused onto the surface of a thin film working electrode through a transparent Sapphire window and thin layer of electrolyte (total optical path of 3.25 mm). The electrolyte thickness of 2.25 mm ensures free diffusion of ions (e.g. protons) and its counter ions.
The electrode adapter for installing 6 mm dia. electrodes inside Raman ECFC is available here. It enables using disk electrode, plug with clip or any other rod-shape electrode (e.g. graphite rod) or current collector (e.g. metal mesh, graphite coated metal mesh, metal foil, graphite coated metal foil, metal foam, carbon woven and non-woven fabrics, carbon paper, etc.) as working electrode instead of a flat electrode in default Raman ECFC configuration.
The screw mount variant of Raman ECFC can be converted into GDE Raman ECFC by using Gas Compartment. This conversion allows installation of Gas Diffusion Electrodes as working electrode.
The cell elements are constructed with materials that are inert to the sample (PEEK, Fluorocarbons). It well fits aqueous (FKM O-Rings) and organic solvent (FFKM O-Rings) electrolyte requirements. The construction is gas-tight and can be used when the removal and exclusion of contaminants such as oxygen and water is required by bubbling of an inert gas through the electrolyte (in an external reservoir).
Application note:
This cell can be used to track kinetic phenomena such as the near-surface proton concentration changes during oxidation and reduction reaction at working electrode. It can be also used to identify materials such as carbon, metal oxides, polymers and electrolytes, and to determine their structure and distribution. Various metals are suitable for this cell as auxiliary electrode including Platinum, Gold and Silver.Specification:
nominal exposure area: 3.5 cm2
electrolyte volume: 4.5 mL
optical path (including Sapphire window): 3.25 mm
electrode plug diameter: 6 mmIntrastat data:
HS Code: 90275000
Country of Origin: Sweden
NET weight: 200 gProduct includes:
1 x chamber
1 x Reference electrode (Ag/AgCl, or Ag/Ag+), 30mm
1 x Metal wire auxiliary electrode – ST 0.6/150 mm, Platinum
1 x Sapphire window – 0054PSPCM
1 x lid
1 x WE Tantalum contact
1 x sample holder
1 x plug – 0005CPEMA











